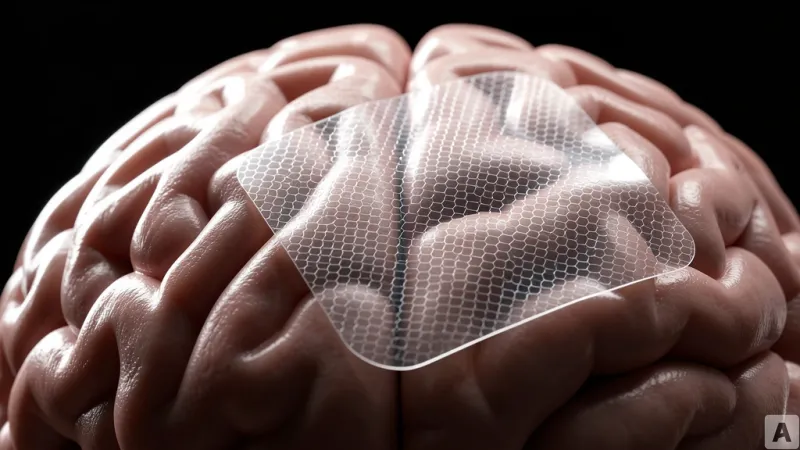
Under the sterile glow of an operating theater, a neurosurgeon navigates the delicate landscape of a patient's brain to excise a tumor. Instead of relying solely on the traditional, rigid metal electrodes that have defined neurosurgery for decades, the surgical team places a gossamer-thin, flexible film directly onto the cortical surface. This material, composed of graphene, captures high-fidelity electrical signals from individual neurons in real-time. By integrating this data, the surgeons can map language centers with unprecedented precision, ensuring that the tumor is removed without compromising the patient's ability to speak. This scene marks a definitive shift: brain-computer interface technology has officially migrated from the controlled environment of the laboratory into the high-stakes reality of clinical surgery.
Clinical Validation and Surgical Integration
Inbrain Neuroelectronics, a company specializing in graphene-based neural signal decoding, has reached a critical milestone by completing the recruitment phase for its first human clinical trial. The study, which enrolled 10 patients, successfully performed surgical procedures on eight individuals, yielding a comprehensive dataset for the research team. This landmark trial is officially registered under the identifier NCT06368310. The project is sponsored by the University of Manchester and conducted in collaboration with the Northern Care Alliance NHS Foundation Trust, a major public healthcare provider in the United Kingdom.
The primary objective of this study is to establish the safety profile of the graphene cortical interface within the context of brain tumor resection. Beyond safety, the researchers are rigorously evaluating the signal quality, long-term stability, and stimulation capabilities of the graphene array. Crucially, the trial also assesses the device's compatibility with existing surgical workflows, ensuring that the integration of this advanced material does not disrupt the standard operating procedures required for complex neurosurgical interventions.
The Material Shift in Neural Mapping
For years, the gold standard for measuring brain activity has been the use of rigid, metallic electrode arrays. While effective, these tools suffer from a fundamental physical limitation: their lack of flexibility prevents them from conforming to the complex, irregular topography of the human brain. This mismatch often results in signal degradation and a loss of spatial resolution. Inbrain’s graphene electrodes solve this by utilizing a material that is only micrometers thick and inherently flexible. This allows the sensor to drape perfectly over the cortical surface, maximizing contact and significantly increasing the signal-to-noise ratio.
During the trial, the graphene interface was used during awake craniotomies, where patients are kept conscious to perform language tasks while surgeons map functional areas. The graphene sensors integrated seamlessly with existing monitoring systems, capturing high-resolution neural signals that were previously difficult to isolate. Perhaps most importantly, the clinical reports indicate no device failures or adverse side effects throughout the surgical process or the immediate post-operative period. The research team is currently in the phase of evaluating primary endpoints, which include a 90-day safety monitoring period and follow-up neuroimaging to ensure the long-term integrity of the cortical tissue.
This successful clinical application confirms that material innovation in brain-computer interfaces has moved beyond theoretical potential to deliver tangible medical value. As the final results are prepared for publication later this year, the data will likely serve as a benchmark for the next generation of medical devices that require high-precision neural decoding.