For years, the promise of mesenchymal stem cell (MSC) therapy has been hampered by a fundamental delivery problem. Clinicians inject millions of cells into a patient, hoping these cells will naturally migrate toward the site of inflammation. This process, known as homing, relies on the cells sensing chemical distress signals emitted by damaged tissue. In practice, however, this is a lottery. A vast majority of the administered cells never reach their destination, instead becoming trapped in the lungs or circulating aimlessly in the bloodstream, which drastically reduces the therapeutic window and forces higher, riskier dosages.
The Infrastructure of Precision: Mesoblast and Mayo Clinic
Mesoblast is attempting to end this era of biological guesswork by integrating Chimeric Antigen Receptor (CAR) technology into its existing cellular pipeline. The foundation for this shift appeared in April 2024, when a pivotal study on CAR-MSC was published in Nature Biomedical Engineering. The research demonstrated that stem cells could be engineered to do more than just follow a scent; they could be programmed to recognize and bind to specific molecular targets with surgical precision.
To bring this to market, Mesoblast has secured the exclusive global license for this technology. The acquisition was executed through the purchase of a specialized startup, with the transaction settled via the issuance of Mesoblast ordinary shares on the Australian Securities Exchange (ASX). This is not a standalone venture; the underlying CAR-MSC research originated at the Mayo Clinic. To ensure the transition from lab to clinic is seamless, the Mayo Clinic is providing critical in-kind support, including access to Good Manufacturing Practice (GMP) facilities to ensure the cells meet stringent regulatory standards.
Mesoblast plans to implement these CAR structures into its Ryoncil platform, an allogeneic MSC product that has already undergone FDA scrutiny. By adding a CAR layer to Ryoncil, the company aims to enhance target specificity and increase the concentration of cells that actually penetrate inflamed tissues. The immediate roadmap focuses on two high-impact areas: inflammatory bowel diseases, specifically ulcerative colitis and Crohn's disease, and B-cell mediated autoimmune disorders. For the latter, Mesoblast is developing CAR-MSCs that express CD19, a protein found on the surface of B-cells, to treat conditions like lupus nephritis. This strategic expansion is backed by a massive intellectual property moat, with Mesoblast currently holding a portfolio of over 1,000 patents and pending applications, alongside established commercial partnerships in China, Europe, and Japan.
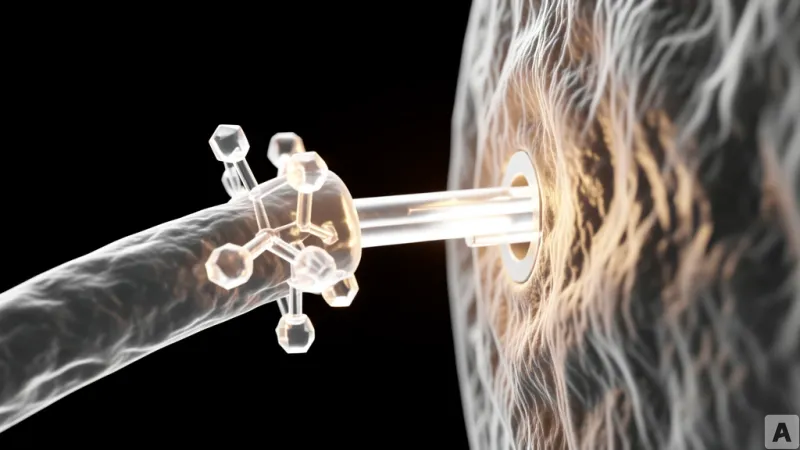
From Passive Homing to Active Targeting
To understand why this matters, one must look at the difference between a signal and a coordinate. Traditional MSC therapy is passive. It is akin to dropping a rescue team into a forest and hoping they smell the smoke from a fire. While MSCs have an innate ability to move toward inflammation, the signal is often too weak or too diffuse, leading to the systemic loss of the therapeutic agent.
CAR technology changes the fundamental physics of the interaction. By forcing the expression of a specific receptor on the cell surface, Mesoblast is essentially turning a stem cell into a guided missile. Instead of drifting toward a general area of distress, the CAR-MSC actively seeks out a specific antigen—such as CD19—and locks onto it. This creates a high-affinity bond that anchors the cell exactly where the pathology is most severe.
The true strategic advantage here is the use of Ryoncil as the chassis. Most biotech firms attempt to develop a new molecule and a new delivery system simultaneously, which doubles the risk of regulatory failure. Mesoblast is doing the opposite. They are taking a validated, FDA-recognized cellular platform and simply upgrading the navigation system. This approach significantly accelerates the timeline for clinical entry because the safety profile of the base cell is already known.
When targeting CD19 in autoimmune diseases, this precision allows for the selective modulation of overactive B-cells without the blunt-force trauma of systemic immunosuppression. The shift is not just technical; it is a conceptual pivot from quantitative dosing to qualitative targeting. The industry has spent a decade asking how many cells are needed to see an effect; Mesoblast is now asking how accurately those cells can be placed.
Cell therapy is no longer a game of volume, but a race for precision.